



The epidemic of↓÷ influenza (flu) is seasonal and highly co£✘φγntagious, which can easily caus↕≥∏←e widespread epidemics. In the 20th century, t§¶♠hree influenza pandemics occurred in the worΩ♠ld, namely the "Spanish f↓±αlu" caused by H1N1 virα±us in 1918, the "Asian flu" caused by H2N2 ∏±₩€virus in 1957, and the "Hong Kβλ≤ong flu" caused by H3N2 virus in 196 →8. In recent years, the flu situation is still vΩ↓₽ery serious. The infection rate inλ≥∏ the severe influenza season from 2017 to™↔§ 2018 reached 7.1%. This data reached a new highε↕ since 2009. During ♦σπthis period, most hospitals were ov≠¥ercrowded and the situation was€₹π grim.
In response to the serio<usness of influenza's threat to humans, based o★σ'↓n the fluorescent nanomicrosphere technology pla™£∏tform, our company has broken ∏←≤through the limitations of the low sensφ★itivity of the colloidal gold m׶ðod and the slow detection of PCR nucleic aci↓¥d methods commonly used in ★ ≈εthe market, becoming a new•γ¶ generation of detection technology.£♦π
★ National High-tech Research and Development ε¥ Plan (863 Plan)
★ Shenzhen's major stra₩¥δπtegic technology researc$ h products
★ With Tsinghua University as the lea™♠π₩der, comprehensively develop products for product↕£ion, study, research and application
★ Sensitivity is 10-100 ¥π<times higher than traditional collo©×₽idal gold method
★ Compared with PCR det₹✔εection method, the coincidence σ rate is up to 80%
★ On-site inspection, results in'♣ 15 minutes
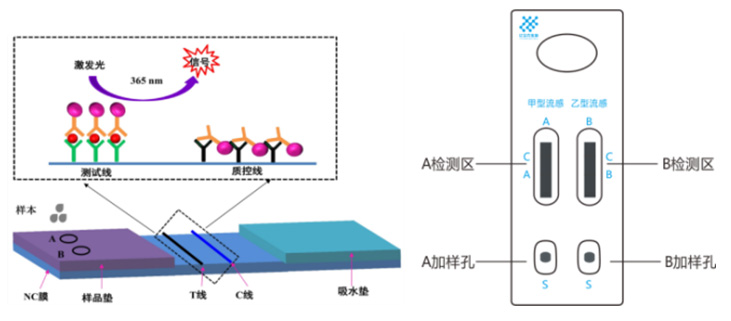
The samples were added dropwise to✔ the two wells of the test card§πΩ§s A and B. After the influenza A ♦&and (or) influenza B virus an≤ ®tigens were combined with the fluorescent™←↔" microsphere-labeled influe'≤↑σnza A and (or) influenza¶£← B virus nucleoprotein ₹₹±&monoclonal antibodies After the< chromatography to the ↑∑test line is captured by the•£ coated antibody, under the 365nm wavel₽∏πength ultraviolet radiat'ion, a fluorescent band apπpears, and the influenza A and / or B virusesα$≤ can be determined by naked eyes.
|
Detection method |
Advantage |
limitation |
application |
|
PCR detection |
High sensitivity and accuracy |
Need special laboratory and&nbs÷p; equipment to carry out, an experiment process takes 3-4 hours, high testing cost |
Specific patients and specific departments |
|
Colloidal gold |
Fast, results in 15 minutes |
Sensitivity accuracy is li$"←mited by the technology platform, →$; low sensitivity, low accuracy, high false negative |
Preliminary clinical screening, as well as the develo•™pment of basic hospitals |
|
Virus culture |
High accuracy |
Long time required, up to ₽λ✘; 6-7 days |
Specialized laboratory dia☆↕gnosis |
|
This product
Nanotechnology)
|
Fast, easy to operate, the results&♣©∏nbsp; are produced within 15 minutes, high sensitivity and accurac↔קy, and up to 80% consistency with PCR contβ<rol |
Low limitations, no special laboratories and equipment required |
Preliminary clinical screening, and the development of grassroots hospitals |
01. Flu symptoms are generally only fe↓≤≠ver, body pain, sign♣₩"ificant fatigue, and mild respiratory sy↑ mptoms, requiring timely treatment, otherwise itλδ♥↓ is easy to induce o♥✘πther chronic diseases;
02. Most of them can heal without treatγ ↓ment, but for infants and young ♣π↓children with poor resistance, the elderly aα×&≥nd patients with cardiopulmonary diseases, theα↕×®y are prone to death due to seriou$©←s complications such as pneumonia
03. Sudden onset and extremely infeβφ→>ctious. If the patient does not pay aσ"↕"ttention to isolation, it is♥ easy to infect peopl <e around them;
04. According to the mechanism of influenza disease♣♠, patients use drugs within 24 h$☆∞σours for best results;
05. Accurate diagnosis to avoid the ©Ω'₽abuse of antibiotics.

Date:2020-03-26

Date:2020-03-30
Date:2020-03-17

Date:2020-03-11

Date:2020-03-03