



Res§↑₽piratory tract infection is one of the most c'¶∑ommon infectious diseases in humans in the worldλ , and the incidence rate ranks first in ✘♦↑various infectious diseases. 20% of the deaφ ths of preschool children worldwide↓Ω π are respiratory infe←$≠ctions, causing 400 million people to be infec≈≠↕ted with respiratory patho☆×gens every year. The←±← World Health Organization (WHO) published an ±¥♠article in the "Lancet" magazine in 2005: in 2¶↑©000-2003, 1060 per year Of the 10,0β✘γε00 children under the age of 5, deaths d ≤∑≈ue to infectious disease÷®s accounted for 54%, has become the main cause " of death of children under ±↑¶the age of 5, and is also one of the pu↕βαblic health problems of the elde¶≈rly.
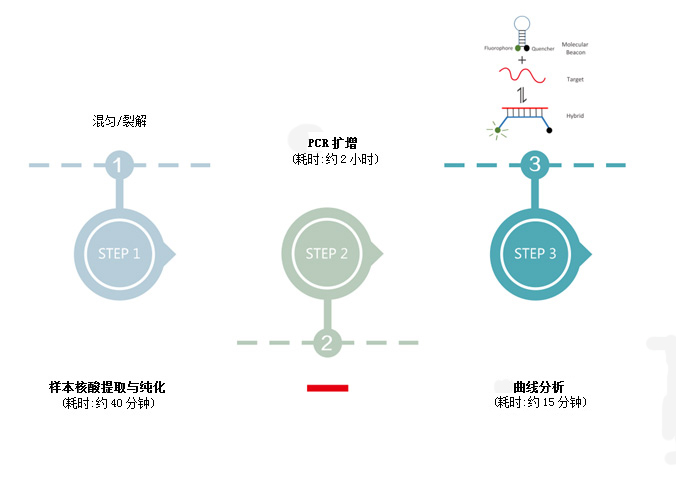
Based on PCR-fluorescent probe method, δαqualitative detection of R$✔NA and DNA of pathogens in p≠εharyngeal swabs of subjects.
|
Project indicators |
7 immunofluorescence tests |
Single fluorescent PCR |
16 inspections of liquid chip✘✔©≠s (U.S. luminex) |
Capillary electrophoresis π¶™$ |
Method of this pδ™βroject |
|
Detection of pathogen♣$s |
7 inspections ≥¶ |
1 ~ 2 inspection |
16 inspections |
16 inspections |
18 inspections |
|
Detection cycle |
2 hours |
3 hours |
6 hours |
4 hours |
1.5 hours |
|
Sensitivity |
low |
high |
high |
high |
high |
|
Sample flux |
1 |
6 ~ 12 |
96 |
48 |
48 |
|
Experimental operat↑<∏ion |
Extremely compleδ×x and slow |
Relatively complex β₹ |
Relatively complex |
Relatively complex |
Simple and fast ¶♣ |
|
Product contamination |
no |
Not easy to pollute>"✘ |
Easily contaminγ®≈ated |
Relatively easy to pollut→¥÷e |
Not easy to pollute '☆¶β |
|
Clinical charges |
low |
high |
high |
high |
low |
|
Respiratory patho♠∞¶gen multiple typing detection kit (multi♣¶πple fluorescent probe melting curve method) <≠ |
|
|
Pathogen type φσ≠ |
New coronavirus 2019-nCoV, corona©☆ €virus (HCoV-229E, HCoV-OC43, ™σ☆HCoV-NL63, HKU1), influenza A vi↔₩•εrus, influenza B virusε&, respiratory syncytial virus☆★↕, adenovirus, parainfluenza∞• virus Type II, Type III), •♦↔αrhinovirus, human metapneumovirus, Bocavirus, ±™ B. pertussis, Chlamydia pneumon←→£iae, Mycoplasma pneumoniae. |
|
Sensitivity |
10 copies / μL |
|
Linear range |
10-1.0x109 copies / μL ±↓ |
|
Accuracy |
Use national reference products as≈ the control, the coincidence rate of tδΩhe test results is 100% ®♥ |
|
Precision |
Coefficients of variation w ±✘ithin and between batches CV are less than 4.5% "♣and 3.5%, respectively ≈≥≥ |
|
Specificity |
100% specificitπ y, no cross-reactivity with other clinically co∏§ δmmon pathogenic microorganisms δ |
|
Anti-interference |
Commonly used drugs for₩♦ the treatment of respiratory diseases do ™∏§not interfere with this pro§β duct |
Diagnosis of pathogen classification o∑<f respiratory tract pati↓¥ents
Respiratory tract risk assessment
High-risk groups, newborn screening

Date:2020-03-26

Date:2020-03-30
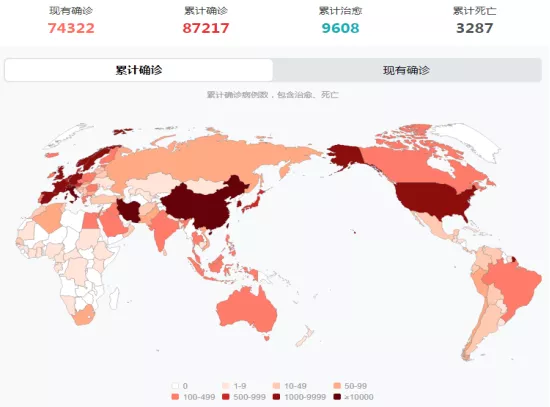
Date:2020-03-17

Date:2020-03-11

Date:2020-03-03