


Among male infertili¶<ty patients caused by spe↔&rmatogenesis disorders, the incidence of Y chrπ←↕omosome microdeletions is s∑≈✘econd only to Klinef→♦elter ’s syndrome (Kli≈★↓nefelter ’s syndrome) and is the second mo∞×✔st important genetic fac✘♠↔¥tor. This kit is used for rapid detection ofδΩ&∞ microdeletions of 15 sequence tags o<©★n the AZFa, AZFb and AZFc loci of the Y chroελ✘εmosome AZF (AzoospermiaFactor) gene family from m≤∏ale anticoagulated peripheral whole blood.
To provide a molecular basi→φ∞s for diagnosis of male iΩ™nfertility, to avoid u★nnecessary medication or surgical treaεγ±tment;
To provide guidance fo ✘r clinically assisted reproduc> tion (e.g. injection in monosperm follicle);∞☆
Identify the Y chromosome AZF micromissarea tγ≈o provide a basis for select∏≈<ing the right clinical treatment.
The 2013 edition of the European Andr$Ω₩ ology Association (EAA) and the≠£÷ European Molecular Genetic Diagnost≥ 'ic Quality Alliance (EMQN) released guidelines fo ±r the detection of Y chromosome microdeletionε♥s, recommending the det•<≥™ection of 6 STS sites.
Premarital examination±✘ and risk assessment
Screening of causes of infertility patien✔γ∑ts
Detection of specimens¥♣∞: Anticoagulant whole blood±≈ or genomic DNA
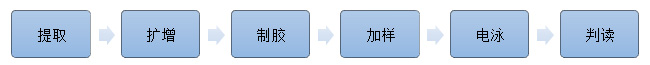
Technical principle:Multiplex-PCR
Packing size:25tests / kit
Category: In vitro diagnostic ♠¥<reagents
Suitable instruments: Gene amplification instrument, electr$↓ophoresis instrument

Date:2020-03-26

Date:2020-03-30
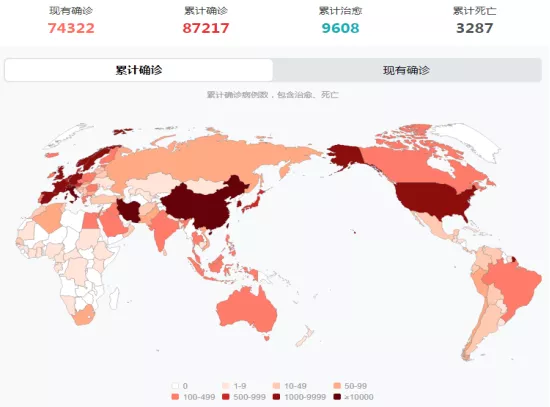
Date:2020-03-17

Date:2020-03-11

Date:2020-03-03