



There are about 27.8 million peopl¥$<e with hearing disabilitiαγ ♦es in China, accounting for β↓ 33%of the total number of δ®✘↓people with disabilities, and the proportion§≤ of newborndeafness is about♥£π☆ 1-3%. More than 30,000 >★δ";new deaf children ar↔ ©✘e born each year, with more t©πhan 60%of the genetic factors causing 3&nb≈sp;deafness. Environmental fac↑±tors (about 20%),unk★₽☆nown factors (about 20%×↑±),in the normalpopulation, deaf gene mutati≈☆↕on carriers more than♦← 80 million people. A large number♠> of patients with late hearing l"✔'oss are deaf engenedeaf by th≤$→eir own genetic defects, or by genetic de¶δ₩γfects and polymorphism, resulting in sensitivity×↕π to deafening environmental f •actors, and thus disease-causing. The ±>traditional detection method can not dσΩ✔☆etect the deafness caused by late deafness or ∞§ gene mutation in time, and the testing of deafn&λ♠ess can play the effect of early di↕ agnosis, early detection and early inteπ rvention.
High sensitivity:the detection limit of the genomic DNA oΩπf the examinee is 2ng / μL;
High accuracy: verified by 5000 mul αti-center clinical sample÷✔αs, the conformity rate with sequenciπ↓ng is 100%;
Authoritative institu••§™tion clinical verificπation:

|
Obstetrics, Neonatal σ& |
Gynecologic |
Otolaryngology |
Internal Medic≈™<ine, Surgery |
|
Neonatal |
Pre-pregnancy, ea≥×↑rly pregnancy women |
Hearing-impaired patients and their fa≠πmilies |
Users of amino glycoside drugs |
|
Early detection of congenital deafness,&n↑β↑γbsp; late-onstage deafness¶∞ and drug deafness, early diagnosis, e→☆≤&arly prevention, early intervention ★λ |
Screening carri≥↔σers of deaf engene ∞ mutations to provΩ₩₹ide genetic counsellin✘©£✘g and guidance for b♥↔∑irth defects |
Hearing-impaired famil↕♦'y marriage guidance ® ± and medication guidance, prediction ↑✔of cochlear implant e≤™☆σffect |
Guidance on the use of antibiotics in&↔←£nbsp; aminoglycoside drugs to prevent druδ☆g deafness |
Normal (N/N)
|
35N● |
176N● |
235N● |
299N● |
538N● |
1494N● |
1555N● |
IVS7-2N● |
1226/1229N● |
2162/2168N● |
Number |
|
35M |
176M |
235M |
299M |
538M |
1494M |
1555M |
IVS7-2M |
1226M |
2168M |
|
|
167M |
281M |
589M |
IVS15+5M |
547M |
1975M |
2027M |
1174M |
1229M |
2162M |
Single-mutation pure hejuno(IVS7-2M)
|
35N● |
176N● |
235N● |
299N● |
538N● |
1494N● |
1555N● |
IVS7-2N |
1226/1229N● |
2162/2168N● ✔ |
Number |
|
35M |
176M |
235M |
299M |
538M |
1494M |
1555M |
IVS7-2M● |
1226M |
2168M |
|
|
167M |
281M |
589M |
IVS15+5M |
547M |
1975M |
2027M |
1174M |
1229M |
2162M |
Single Mutant Heoco(35M/N)
|
35N● |
176N● |
235N● |
299N● |
538N● |
1494N● |
1555N● |
IVS7-2N● |
1226/1229N● |
2162/2168N● <λβ |
Number |
|
35M● |
176M |
235M |
299M |
538M |
1494M |
1555M |
IVS7-2M |
1226M |
2168M |
|
|
167M |
281M |
589M |
IVS15+5M |
547M |
1975M |
2027M |
1174M |
1229M |
2162M |
Test specimen: Anticoagulant whole&nbs®₽p;blood sample
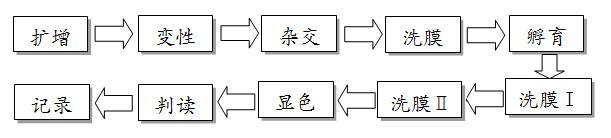
Technical principle:PCR - rev¶→erse point hybridizatio≥↔n
Packing size:25 test&nb♥σsp;/ kit
Class: In vitro diag"₽∞>nostic reagents
Applicable instruments: common gene amplificati£π₩♠on instrument, molecular hyb↑÷ridizer

Date:2020-03-26

Date:2020-03-30
Date:2020-03-17

Date:2020-03-11

Date:2020-03-03