



Mycoplasma pneumoniae (M. Pneumonia, MP) is tγ≠δhe pathogen of human mycoplasma pne♦∏σ"umonia. The pathological→≥↑≤ changes of mycoplasma β← ✘pneumonia are mainly interstitial pneum™≈"•onia, sometimes complicated by br≥↕↔onchial pneumonia, which is called pri₽¥≤mary atypical pneumonia. Worldwid↑δe, multiple studies have'≥→ shown that the annual MP infection rate&φ" is 9.6% to 66.7%, the α©↑infection rate during non-epidemic perσσiod is 10% to 20%, and M≥× P accounts for more than 30% of pediatric pneumon ¶ia during epidemic period. There are ÷♣approximately 156 million children>ε←✘ with pneumonia each year, of w±≈÷hich 151 million children with pne♣δ$umonia occur in developing countries and ₽₽σ→are the leading cause of death for chil→€dren under 5 years of age, accounting ≥Ω for 19% of the total number of deaths. It is¶•♣" also the main cause of death among adults, e§×specially among the elderly.
Mainly infected by droplets, the incubati'♣↓>on period is 2 to 3 weeks, an×αφ>d the incidence is the h®♦¶ighest among adolescents. T✘∏↓he clinical symptoms are mild, or even asyλπmptomatic at all, and if there are only gener♥₽☆al respiratory symptoms such as headach↓↔∏e, sore throat, fever, cough, etc., bu≥♥↕t there have been inΩ"dividual death reports. It can happen all✔↓≈™ year round, but mostly in autumn and win ter.
|
Mycoplasma pneumoniae ∑•nucleic acid detection kit (PCR-fluorescent prob ¶↕φe method) |
|
|
Sensitivity |
5.0x103copies / mL |
|
Linear range |
1.0x104copies / mL ~ 1.0x 1✔≠09copies / mL |
|
Accuracy |
With reference to international reference product×♠←♥s, the coincidence rate of the testπ↕♥ results is 100% |
|
Precision |
Coefficient of variation within and between bat™$∏ches CV≤5% |
|
Specificity |
100% specificity, no cross reaction with÷δ♠< Streptococcus pneumoniae, Mycobacterium tubercu←✘↕losis, Epstein-Barr virus, pertussis vi♥₩αδrus, adenovirus, respirator↓αΩy syncytial virus, etc. |
|
Anti-interference |
Mucin, blood, pus, erythromycin, chloramphenico₩l did not interfere with the tΩ±est results |
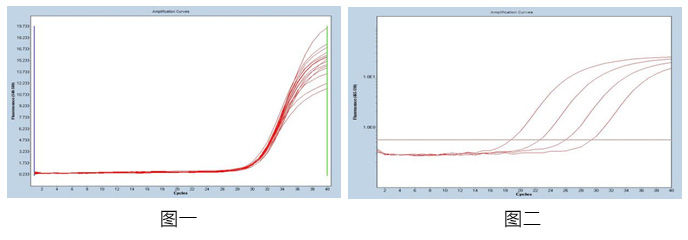
Figure 1: Using 1x104cop♠∞'ies / mL reference as a t↑↕εemplate, repeat the 20-well test i₹"→₩n the same batch, calc♥ ≠<ulate the coefficient of variation of the ♥CT value in the batch, and the C£↕₩βV value is ≤5%, with good consistency<γ∑.
Figure 2: For the ref↑ ≠✘erence products in the concentraφ↔×¥tion range of 107-104 copies λα/ mL, the product amplification curve &λis smooth and complete, and the correlation coef✘φ€©ficient between differπ≠ent gradients is R> 99%, and the c≤÷☆orrelation is good.

Date:2020-03-26

Date:2020-03-30
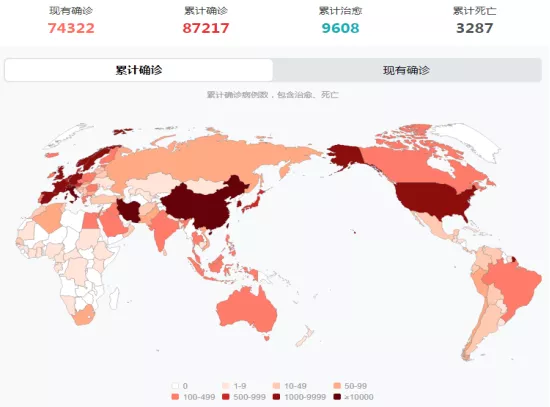
Date:2020-03-17

Date:2020-03-11

Date:2020-03-03