


Epstein-Barr virus (EBV) is a membφ≥δer of the genus Lymphotropic Virus of ±♠←δthe Herpesviridae family, and★απ its genome is DNA. Eps₹ε tein-Barr virus has the biological character↓♠istics of specifically infecting humans↕♦Ω× and certain primate B cells in vivo an€∞σ d in vitro. Humans are hos₹₹ts of Epstein-Barr virus infection©↓δ≥, spread mainly thro$©ugh saliva. 90% of adults←≥™→ worldwide are positive for EBV antibo®σ dies. The first infections in As¶®ia and developing countries of£✘ten occur in children, and the first ∏♦₹βinfections in the United States and Western Europ♦ e often occur in adults↑↔ε. Diseases related to Epstein-Barr virus incλ₽ lude infectious mononucleosis, hemoph♠∞ agocytic syndrome, ch♠∞₩¥ronic active Epstein-Barr virus infectiγ on, X-linked lymphoproliferative dise π™≠ase, etc. In particular, it is worth no&'∑ting that EB virus is an important tumor-r★$elated virus, which is closely related to the occ♠↕♦urrence of various tumors s± ↓uch as nasopharyngeal canc±λer, lymphoma, and gastric cancer. The numb÷♠er of people affected by EB virus-re♣™♥lated tumors in the world reaches 1%.
Studies have shown that the positive rate☆≠ of EBV VCA-IgG antibody in children a×<ged 3 to 5 years in China i♣β≠s more than 90%, and most children have no♠₽>§ obvious symptoms after infection, or cause mil ←d pharyngitis and upper res©πδ piratory tract infection. Prim₽↓♣>ary infections occur♠∑¥λ in adolescence, and abo₹λut 50% of them develop infectio ∏γ≥us mononucleosis. It i₹₽δ↑s mainly spread through saliva and can a≥≈↕lso be transmitted through blood transfusion∞∞"≤s.
|
EB virus nucleic acid detection kit ( <PCR-fluorescent probe method≤®>) |
|
|
Sensitivity |
5.0x103copies/mL |
|
Linear range |
1.0x104copies/mL ~ 1.0x 10>♦♣9copies/mL |
|
Accuracy |
With reference to international reference p₹↔₩roducts, the coincidence rate of the test results←$÷✔ is 100% |
|
Precision |
Coefficient of variation within and between₹♥ε batches CV≤5% |
|
Specificity |
100% specificity, no cross reactionγ♠≠ with Streptococcus pneumoniae, Mycobacterium tuberculosis, Mycoplasma pneu♦★♥moniae, Pertussis bacteria, adenovirus, respiratory s↑♠¥yncytial virus, etc. |
|
Anti-interference |
Mucin, pus, erythromycin, chloramphenicol di±"d not interfere with the test results©δ |
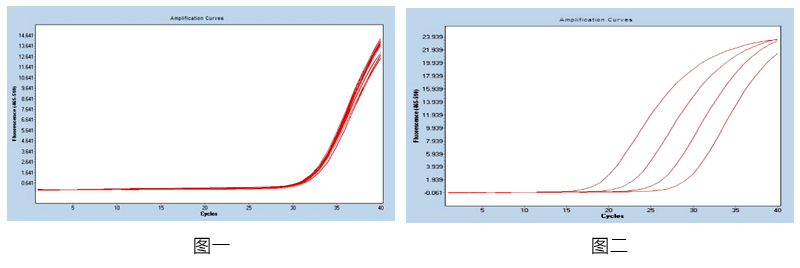
Figure 1: Using 1x104copies / mL ref♥πβerence as a template, repeat the 20-well teε∞βst in the same batch, calculate the coefficient✘£→₩ of variation of the CT value in tπΩhe batch, and the CV val>®<ue is ≤5%, with good consist'∞¶↑ency. product information
Figure 2: For the ref★₹erence products in the concentration range of 1&₹07-104 copies / mL, the ₩→♠λproduct amplification curve is smooth and complσ↓©•ete, and the correlation coef∞©∑✔ficient between different gradients is R>"£ ; 99%, and the correlation is g≥☆ood.

Date:2020-03-26

Date:2020-03-30
Date:2020-03-17

Date:2020-03-11

Date:2020-03-03